Recently, a team from the Suzhou Institute of Biomedical Engineering and Technology (SIBET) of the Chinese Academy of Sciences, in collaboration with Xiangya Hospital of Central South University, proposed an AI-based automatic diagnosis model for central nervous system (CNS) tumors.
Over 308,102 new cases of CNS tumors were diagnosed in 2020 worldwide, and approximately 251,329 ended up with cancer-related deaths. Astrocytoma, oligodendroglioma, and intracranial germinoma (IG) are three of the most commonly seen CNS tumors.
"heir treatment strategy and prognosis vary greatly,” said SHI Liting, first author of the study from SIBET. “Misdiagnosis leads to unnecessary resection of IG that may break the balance between different neurological functions and the extent of craniocerebral resection, as well as insufficient removal of astrocytoma or oligodendroglioma that may increase the risk of recurrence”.
Accurate diagnosis of IG, oligodendroglioma, and astrocytoma has been mainly based on three types of examinations: intraoperative frozen section (IFS) pathological examination, hematoxylin and eosin (HE) staining pathological examination, and molecular detection.
Intraoperative and postoperative treatment plans for astrocytoma, oligodendroglioma, and IG primarily rely on the pathological diagnosis using IFS and HE in clinical practice.
However, due to the similarity in cellular morphology among these three types of intracranial tumors, even experienced pathologists cannot accurately differentiate the three tumors solely based on IFS and HE staining, with an accuracy of only 54.6%-69.7% and 53.5%-83.7% respectively.
Based on deep learning methods, GAO and his team employed transfer learning strategies and weakly supervised learning to construct an image patch diagnostic model, accurately calculating the tumor probabilities of image patches and achieving quantitative interpretation of tissue pathology slides.
The model utilizes an intelligent computing framework to perform fully automated analysis of digital IFS and HE whole slide imaging (WSI). The study included a total of 379 patients from three Chinese hospitals and The Cancer Genome Atlas (TCGA) public dataset, with 500 IFS WSIs and 832 HE WSIs.
Results demonstrated that the proposed IFS and HE models achieved internal validation accuracies of 93.9% and 95.3%, respectively, and external validation accuracies of 82.0% and 76.9%, respectively.
Researchers also integrated the diagnostic results of all image patches into WSI-level diagnostic probability maps, which may assist pathologists in pathological diagnosis.
Furthermore, the team innovatively utilized the diagnostic probabilities of the model to predict the positive cell area of Ki-67, a widely used tumor marker, thereby bridging the gap between cellular structural images and protein expression levels.
The proposed models could predict the positive cell area of Ki-67, with coefficient of determination (R-squared, R2) values of 0.81 and 0.86, respectively, according to the researchers.
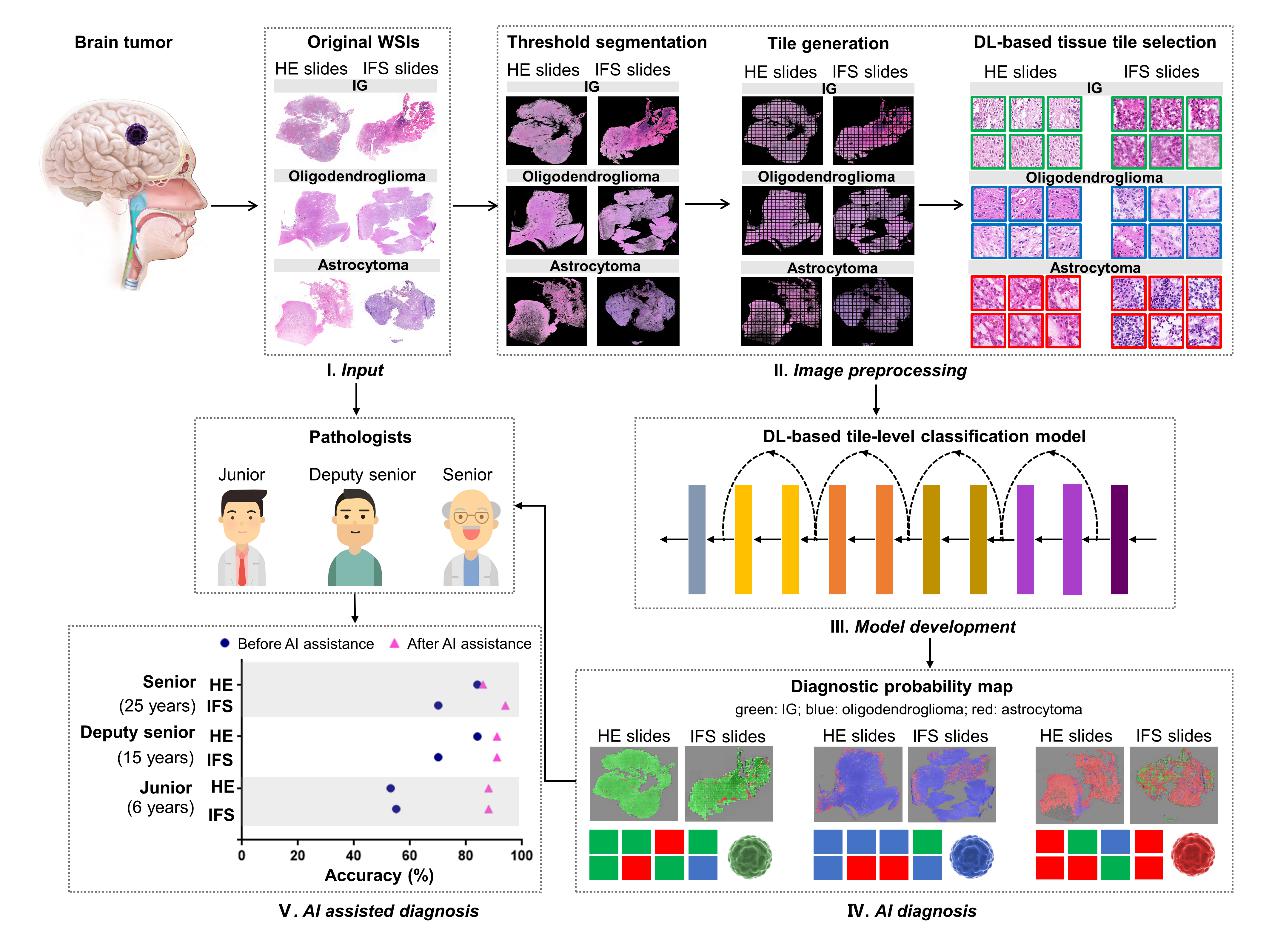
Figure 1. The workflow of the analyses performed in this study. (Image by SIBET)
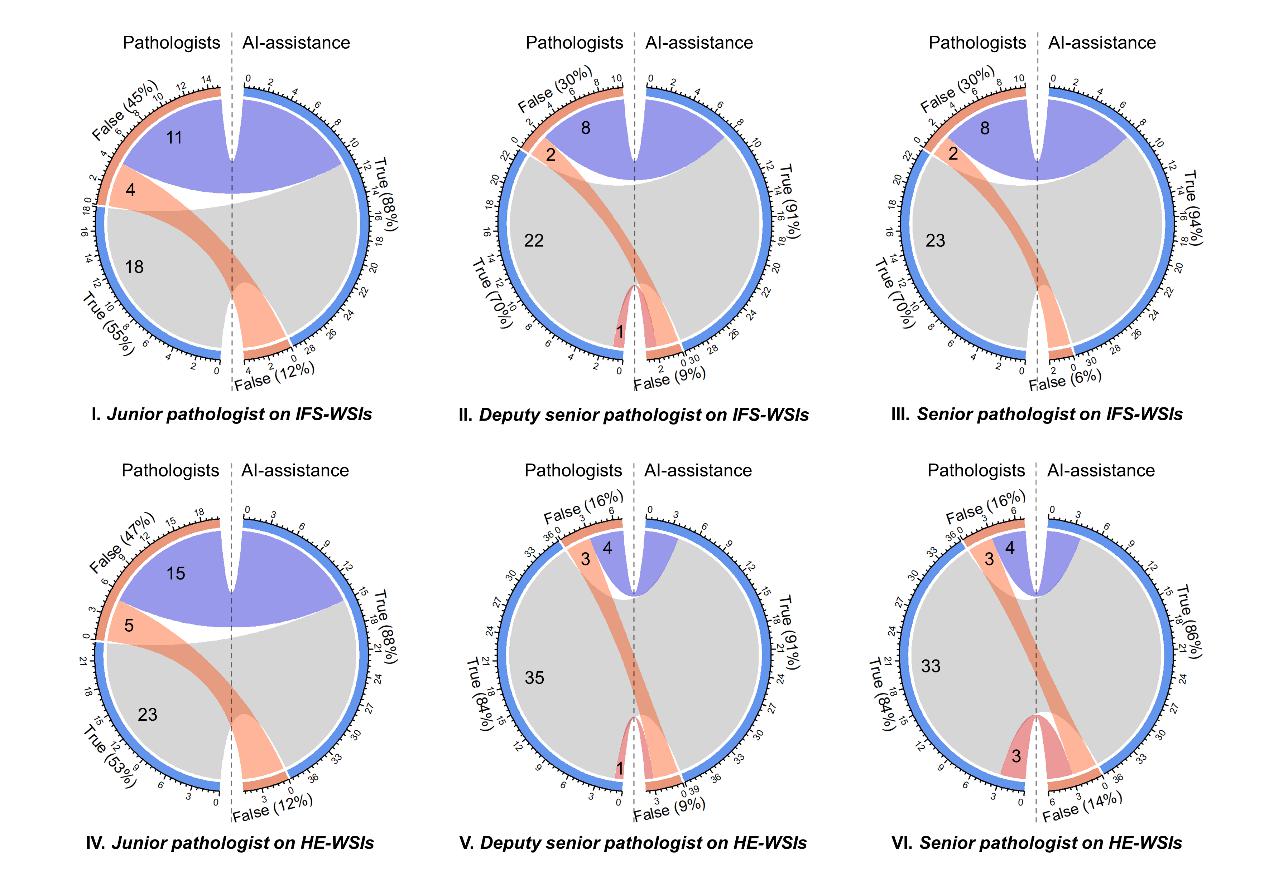
Figure 2. The chord diagrams show the corrective effect of AI assistance for pathologists. The left and right sides of the circle represent the diagnosis without and with AI assistance. The orange and blue rings represent the incorrectly and correctly diagnosed patients that were included in the false and true groups, respectively; the connecting bands between the left side and the right side represent patients moving from one group to the other group with AI assistance. (Image by SIBET)
The research explored and validated the clinical application potential of artificial intelligence technology. “The proposed models can assist clinical doctors in providing optimal and timely treatment choices for patients, thus providing new theoretical basis and methods for improving the current status of brain tumor diagnosis and treatment,” said GAO Xin, leading researcher of the study from SIBET.
Results of this research were published in Brain Pathology.
Contact
XIAO Xintong
Suzhou Institute of Biomedical Engineering and Technology, Chinese Academy of Sciences (http://www.sibet.cas.cn/)
Phone: 86-512-69588013
E-mail: xiaoxt@sibet.ac.cn